General
Projects
Short Term Plasticity of Synaptic Transmission
|
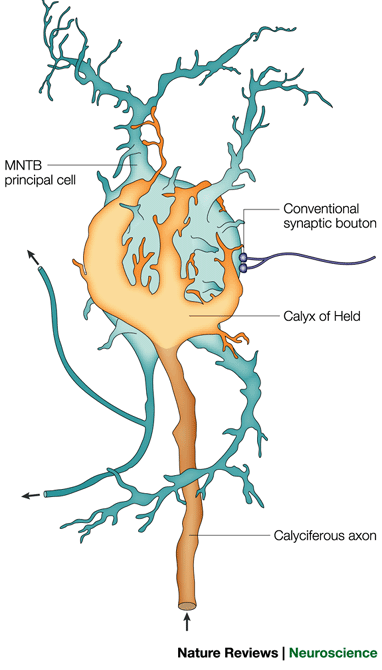
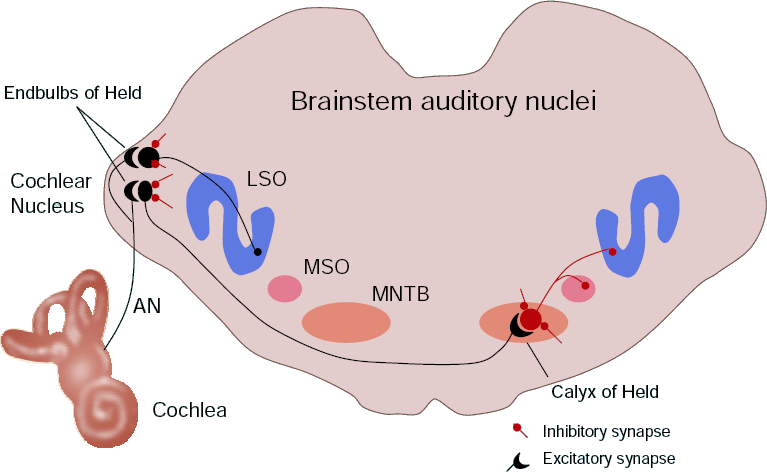
In collaboration with Prof. Ian Forsythe (MRC Toxicology Unit, University of Leicester), we are developing a variety of mathematical models of synaptic transmission at the calyx of Held, a giant synapse in the mammalian auditory system. The aim is to illuminate the pre- and postsynaptic biophysics underlying short-term modulation of the synaptic response. We are using the models to explore the information carrying capacity and signal filtering characteristics of this synapse in an effort to understand its functionality. |

|
|
|
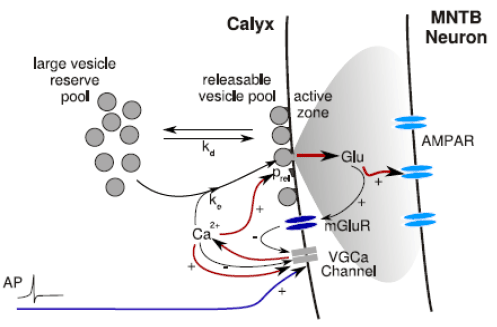
Mathematical Models of the Calyx [top]
A number of variations on the model illustrated above have been developed as an aid to understanding the contributions of different molecular mechanisms (including vesicle recycling and release, calcium channel inactivation, metabotropic glutamate receptor activation and AMPA receptor desensitization) to short term plasticity (depression and facilitation) at the calyx of Held (Hennig et al., 2008; Hennig et al., 2007; Graham et al., 2004; Graham et al., 2001).
These models include stochastic versions that track and release individual vesicles of neurotransmitter (Yang et al., 2009; Wong et al., 2003; Graham et al., 2001) and simpler average response (mean field) models that use deterministic ODEs to represent the average size of the readily releasable vesicle pool and average amount of transmitter release (Hennig et al., 2008; Hennig et al., 2007; Billups et al., 2005; Graham et al., 2004).
The models have sufficiently few parameters that mathematical optimization can be used to match the model with experimental data. Tight fits have been obtained to a variety of experimental data showing depression in EPSC amplitude at various frequencies of stimulation.
Matches to Experimental Data [top]
Detailed modelling of transmitter release and AMPA receptor kinetics predicts that acceleration in EPSCs with increasing temperature may purely be due to acceleration of the AMPA receptor kinetics (Postlethwaite et al., 2007).
The models have also been used to explore the possible effects of presynaptic metabotropic glutamate receptors on EPSC amplitude during depression and recovery (Billups et al., 2005). In line with the data, the models demonstrate that the effects of mGluRs are only observed during recovery from depression.
Experimental data from the Forsythe laboratory indicate that desensitization plays a significant role in short-term depression. They have developed a novel assay for desensitization using low-affinity competitive glutamate antagonists, the efficacy of which has been confirmed by the model (Wong et al., 2003).
Information Transmission and Signal Filtering [top]
We have used the models to calculate the amount of information the postsynaptic EPSC amplitude carries about the previous interspike interval of action potentials arriving at the synapse. We have shown that the EPSC amplitude carries significant information about the presynaptic ISIs even at high (>100Hz) mean frequencies of stimulation (Yang et al., 2009; Graham, 2002). The main information carrying mechanism changes with stimulation frequency.
Short term synaptic plasticity results in a synapse acting as a filter of the incoming spike train. Both depression and particularly the rate of recovery of the EPSC amplitude are stimulus-history-dependent (Hennig et al., 2008). The recovery time course depends on both the loudness and duration of a sound stimulus that generates the incoming spike train.
In a collaboration with Dr Christian Stricker (Neuronal Network Laboratory, John Curtin School of Medical Research, Australian National University), I have investigated the filtering properties of synapses in response to more complex dynamic signals (Graham and Stricker, 2008).
Volume Transmission [top]
|
Synaptic and cellular properties in the MNTB are modulated by volume transmission signals generated through activity-dependent synthesis of nitric oxide (NO). Work in the Forsythe laboratory has demonstrated the NO is produced in the MNTB under physiological conditions and subsequently modulates cell excitability and synaptic efficacy (Steinert et al, 2008). I have modelled the production and diffusion of NO in response to stimulation of MNTB neurons through input to the calyx (Steinert et al, 2008). |
Funding [top]
- 2005-2007
- BBSRC Grant BBS/B/07217, Ł151,551, to B. Graham with a joint grant to I.D. Forsythe (total funding Ł359,495). This grant funded a modelling RA in Stirling and an experimental RA in Leicester to investigate "Changes in information transmission at an auditory synapse in the binaural pathway during short-term synaptic modulation". Read our final report (PDF).
Yang, Z., Hennig, M., Postlethwaite, M., Forsythe, I.D. and Graham, B.P. (2009). Wide-band information transmission at the calyx of Held. Neural Computation, 21:991-1017. (preprint | model code)
Graham, B.P. and Stricker, C. (2008). Short Term Plasticity Provides Temporal Filtering at Chemical Synapses. Artificial Neural Networks - ICANN 2008, LNCS Vol.5164, Springer, Berlin / Heidelberg, pp268-276 (doi:10.1007/978-3-540-87559-8_28)
Steinert, J.R., Kopp-Scheinpflug, C., Baker, C., Challiss, R.A.J., Mistry, R., Haustein, M.D., Griffin, S.J., Tong, H., Graham, B.P. and Forsythe, I.D. (2008) Nitric oxide is a volume transmitter regulating postsynaptic excitability at a glutamatergic synapse. Neuron, 60:642-656 (doi:10.1016/j.neuron.2008.08.025)
Hennig, M., Postlethwaite, M., Forsythe, I.D. and Graham, B.P. (2008). Interactions between multiple sources of short-term plasticity during evoked and spontaneous activity at the rat calyx of Held. Journal of Physiology, 586(13):3129-3146. (doi:10.1113/jphysiol.2008.152124; preprint)
Hennig, M., Postlethwaite, M., Forsythe, I.D. and Graham, B.P. (2007). A biophysical model of short-term plasticity at the calyx of Held. Neurocomputing, 70:1626-1629 (doi:10.1016/j.neucom.2006.10.074)
M. Postlethwaite, M.H. Hennig, J.R. Steinert, B.P. Graham and I.D. Forsythe (2007). Acceleration of AMPA receptor kinetics underlies temperature- dependent changes in synaptic strength at the rat calyx of Held. Journal of Physiology, 579:69-84 (doi:10.1113/jphysiol.2006.123612)
Billups, B., Graham, B., Wong, A., and Forsythe, I. (2005).
Unmasking group III metabotropic glutamate autoreceptor function at excitatory synapses in the rat CNS. Journal of Physiology, 565.3:885--896.
Graham, B.P. (2002). Multiple forms of activity-dependent plasticity enhance information transfer at a dynamic synapse. Artificial Neural Networks - ICANN 2002, Ed. Dorronsoro, J.R., LNCS Vol.2415, Springer-Verlag pp45-50.
Graham, B.P., Wong, A.Y.C. and Forsythe, I.D. (2004). A multi-component model of depression at the calyx of Held. Neurocomputing, 58-60:449-454.